What is Tear Duct Occluder (R) for?
Tear Duct Occluder(R) is a prescription medical device that has to be prescribed by a licensed eye care professional, such as ophthalmologist, optometrist or optician for patients to use with eyedrops. It prevents eyedrops drain through the nasolacrimal ducts into the throat causing drug side effects, ranging from unpleasant bitter taste in the mouth to systemic side effects, such as lowering blood pressure, reduced pulse rate, fatigue, and depression. Some eyedrop medications need prolonged contact time in the eye in order to be effective. This device will keep the eyedrop in your eye so that the medications may infuse into your eye fully and not partially drain away through the tear duct into the throat. Your doctor may have recommended manual nasolacrimal occlusion for you to practice while putting in drops but if it is too burdensome to practice or it is difficult to position your finger right on the nasolacrimal ducts, Tear Duct Occluder is right for you.
This device is primarily for glaucoma patients because glaucoma eyedrops require long contact time in the eye and have relatively serious side effects, if leaking into your throat through tear ducts.
If you use dry-eye eyedrops, such as Xiidra, this device can prevent you getting dysgeusia (bad aftertaste) following your eyedrop instillation. Prolonged using of this device can produce lasting (but not permanent) punctal occlusion effect. So, it is a noninvasive alternative for punctal plugs.
Direction of Use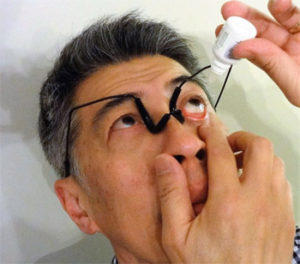
- Put on Tear Duct Occluder, adjust nose pad gaps as needed. Wrap Velcro band around the back of your head and then reduce the band length by 1 cm.
- Pull the lower eyelid down to form a pocket to receive eyedrops.
- Put eyedrops into the eyelid pocket by aiming at the center of the eye.
- Wait for 5 minutes and then remove the device. You may keep your eyes open during this period.
Alternatively, you may put in eyedrops first before putting on the device. However, by doing so the tear duct occlusion would be less effective.
Benefit of Using Tear Duct Occluder
If your doctor advised you that you need to perform manual nasolacrimal occlusion by pressing your tear ducts with fingers while applying eyedrops, Tear Duct OccluderTM offers you an easy and hand-free way to perform the same.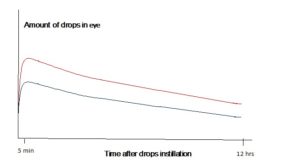
Glaucoma Patients – anti-glaucoma eyedrop efficacy improves if the drops stay on ocular surface long enough to allow full absorption into the eyes. The absorption time curves on the right illustrates the amount of eyedrops medication remains in the eye after the instillation. The lower curve, without using Tear Duct Occluder, shows the drops infuse into the eye in about 2 minutes and the drug effect reaches its peak in about 2 hours and then gradually reduces, giving an average dosage of x% after 12 hours. The upper curve represents higher amount of drops being absorbed into the eyes by using Tear Duct Occluder, giving a higher average dosage, or x+% after 12 hours. Our clinical data shows that with this additional amount of dosages remains in the eye, 83% of our glaucoma patients have lowered their eye pressure by 2 points. Some patients had even postponed surgery.
Dry Eye Patients – if you use Xiidra for your dry eyes and suffered dysgeusia (bitter metallic aftertaste) from the drops, you may use Tear Duct Occluder with the drops to prevent dysgeusia. Our clinical data shows 100% success rate.
There is strong scientific proof that nasolacrimal occlusion following eye drop instillation improves delivery of medication. Prior clinical studies show that the efficacy of applying nasolacrmal occlusion in lowering glaucoma patient’s intraocular pressure (IOP) is about 2 mm Hg. For more detailed safety and effective information, please see Tear Duct Occluder Labeling .
Device Description
Tear Duct OccluderTM is indicated to temporarily occlude the ducts in adult patients to reduce outflow through the nasolacrimal ducts. It is a prescription device and must be prescribed by a licensed eye care professional.
Tear Duct OccluderTM has the “look and feel” of a spectacle frame. It is made up of flexible material that is rigid enough to maintain the shape of the frame during normal use (but not too rigid to hurt the facial tissue) yet soft enough to bend by hand. This material characteristic enables this device be fully adjustable to fit individual patients. The skin contact portions of the frame are covered with silicone rubber (i.e. Latex-free) to improve comfort and increase coverage over the tear ducts.
Safety & Effectiveness
Tear Duct OccluderTM is indicated, as defined by FDA, to temporarily occlude the nasolacrimal ducts in adult patients to reduce outflow through the nasolacrimal ducts. Federal law restricts this device to sale by or on the order of a licensed eye care professional because it requires licensed eyecare professionals to supervise its use. Wear this device for 5 minutes at a time. Patients should consult the licensed eye care professional with regard to the use of this device, in case of bruising after use, open globe, post-surgery, infection, and inflammation. Patients are advised to stop using this device should the skin, in contact with the device, develop irritation. This device should not be used if patients use anti-coagulants.
This device is to be replaced every 12 months. Over-use may lead to device fracture and/or compromising Velcro fastening ability. The device should fit the nasal aspect of patient’s orbital rim seamlessly. Improper fit of the device will render the device non-functional. See troubleshooting guide for details.
Device Adjustments
The nose pads were curved at factory to fit all patients. DO NOT alter the curve or it will became uncomfortable to wear. For someone with wider nose base, two nose pads may be pull apart slightly to accommodate the nose and the adjustment can be done by patients, not by doctors.
Maintenance
Over time, the nose pad sleeves may be coated with some deposit. This layer of deposit may or may not be visible. It is advisable to clean it weekly with an alcohol patch or moistened facial tissue. The service life of this device is expected to be 6 months or when the Velcro fabric becomes loose or shows signs of wear and tear, whichever comes first.
Storage
The Tear Duct OccluderTM should be placed on an open surface or hung on the wall with no objects pressing against it.
Failure time/mode
The Velcro strap of the device typically begins to loosen after 6 months of use. Depending on the number of times the device is used, the failure time may be longer or shorter than 6 months. The failure mode is when the Velcro fabric shows sign of wear and tear.
Disposal
The Tear Duct OccluderTM does not contain hazardous materials and may be disposed as household trash.
Troubleshooting
Feel pressure pain while wearing the device.
Check the Velcro strap tightness. Only a gentle pressure is needed to occlude the tear ducts. Loosen the Velcro strap may solve the problem.
Feel pinch at the corner of the eyes when wearing the device.
This is because the nose pads were set apart too much that reached the corner of the eyes. The remedy is to push both nose pads closer to the nose.
Feel uncomfortable while temples press around the ears.
This problem can be resolved by flip the temple hook upwards.
Temple hook slips out of temple sleeve.
Clean the hook with soap to remove any grease and then reinsert the hook back into the temple sleeve.
Velcro straps pulling woman’s heir.
The pulling of heir is cause by the Velcro loop tape near the temple hook area. The remedy is the wrap some cloth of tape around the area. This problem did not occur on male patients.

