![]()
New Tear Duct Occluder – fits patients universally!
- Improve Eyedrops Absorption
- Prevent Dysgeusia and Other Adverse Systemic Side Effects
- Long term use can provide better and lasting punctal occlusion than using punctal plugs.
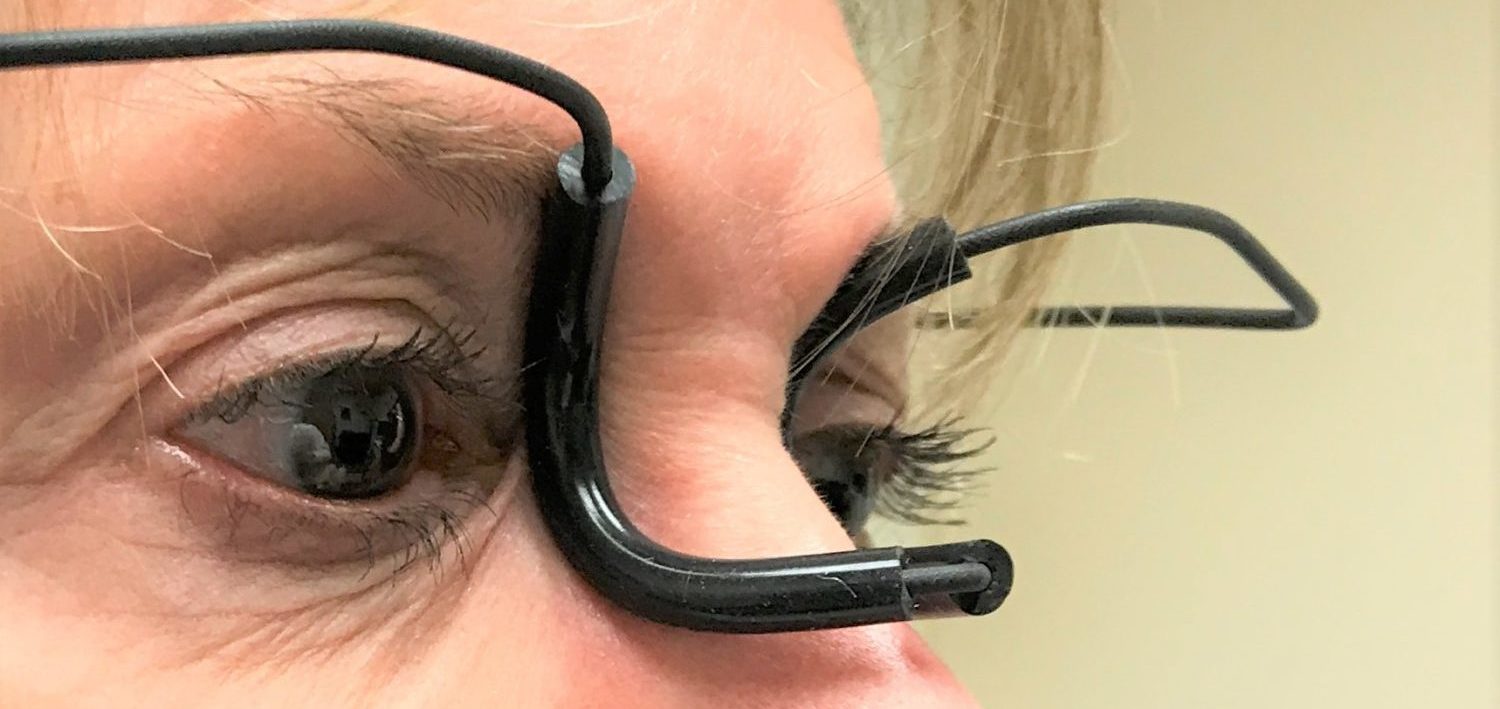
Tear Duct Occluder(R) is an FDA approved Rx Only device which is indicated to temporarily occlude the nasolacrimal ducts in adult patients to help reduce outflow through the nasolacrimal ducts. Clinical studies have shown that to temporarily clude nasolacrimal ducts while putting glaucoma eyedrops can lower IOP further and prevent systemic side effects, such as dysgeusia or lowering pulse rate. Federal law restricts this device to sell by or on the order of a licensed eye care professional. The primary users of this device would be glaucoma and dry-eye patients. See “Physician” Page for more clinical information.
The device has been commercially distributed worldwide since July 2016. The major complaint of the original Tear Duct Occluder was that its nose piece needs extensive adjustments to fit individual patient. The new Tear Duct Occluder does not have this problem! Its nose piece was precisely designed to universally fits all patients. The new design has been tested and proven in worldwide market since 2018. So far, we haven’t received any non-fit complains. Repot from the field shows long term use of this device can produce lasting (but not permanent) punctal occlusion effect. So, it is a noninvasive alternative for punctal plugs.
Here are some of our patients said about Tear Duct Occluder:
I use the device twice daily and I find it has contributed greatly to my continued stability for eye pressure (an average 2 point drop to 17) and comfort in applying the drops with much less to almost no bad aftertaste. It has been a comfortable and stable fit as well. Thank you for such a great device; it has made a difference in many ways. I never want to be without it! Best regards, Barbara North Smithfield, RI.
“I have had two pressure readings since using the device and they seem to be holding at a point or two below my “usual” reading.
I am very happy with the device and truly feel it is working for me! It gives me reassurance that my eye drops are being administered correctly!.” – Kathy, Moultonborough, NH.
“I use it faithfully twice a day since you offered it to me and for the first time my reading on my right eye was 14.5 instead of 16-17 which has been stable the last few months at that reading. It is a drop that we were hoping for…. I would highly recommend your tear duct occluder to anyone with Glaucoma issues. The device also makes it much easier to comply with keeping the maximum efficacy of the eye drops for a full five minutes in the eyes. It’s been a marvelous invention and I truly thank you for it. For me it has delayed the drainage surgery that was my only other option.” – Barbara, North Smithfield, RI.
“It works very well. I do use it 50 percent of the time.” – Michael, Hopkinton, MA.
“I use the device, probably, 80 percent of the time. Sometimes, it is impossible for social or time constraint reasons. I put in (various) drops seven times a day, so you can imagine how difficult it is to always have privacy and time. I can’t be out to dinner and put your occluder on!! One of my eyes is being watched weekly, so I have had many pressure checks over the last two months and my pressures have remained constant. The eye that we are most worried about has not gone up or down.” – Debbie, Medfield, MA.
“It does appear to be working for me. My wife usually puts the Azopt drops in because that one is quite expensive and I hate to miss my eye. It’s comfortable, easy to use and I would recommend it to other patients.” – Tony, Wrentham, MA.
“Been using your device for a couple of weeks while traveling and am very pleased…It’s a wonderful device and convenient for putting in drops and maintaining dosage.” – William, Ashland, MA.
“Pressures are coming down. Very pleased with your device. Very convenient, I do need another one for travel. How and where can I obtain one?
Thanks so much for introducing me to this amazing device. Telling everyone I can about it.” – William. Ashland, MA.
“Xiidra works well for my dry eye but the long lasting dysgeusia from the drops was awful until I used Tear Duct Occluder to prevent the drops from draining into my throat” – Bertha, Weston, MA.
“I used to have a lot of leaks through tear ducts while putting in eyedrops. 4 years ago, I used this device for blocking tear ducts. Now I found that I can do away with this device and still no leak.” – Steve, Weston, MA
Safety and Prescribing information:
click Tear Duct Occluder Labeling 11-9-2022 (also see Device labeling change)
Patent and Regulatory information:
Registered Trademark: Tear Duct Occluder(R)
Federal Regulation 21 CFR 886.5838: Nasolacrimal Comptression Device
Made in U.S.A.![]()